At the forefront of their field, Adena Interventional Pain Management physicians Mark Malinowski and Andrew Porter are now the first in central and southern Ohio to offer their patients the latest solution in treating chronic pain and restoring people’s lives.
The StimRouter by Bioness is the first FDA-cleared minimally-invasive neuromodulation device designed to treat chronic pain of a peripheral nerve origin. The StimRouter can be used to treat pain throughout the body, including, but not limited to:
• Post-surgery pain (knee, back, etc.)
• Post-stroke shoulder pain
• Complex Regional Pain Syndrome (CRPS)
• Amputation pain
• Neuromas
• Neuralgia (excluding cranial facial)
• Carpal Tunnel
“This is the modern way to treat pain,” explained Porter. “The StimRouter gives us the ability to care for a patient who before was untreatable. Up until now, patients suffering from chronic pain would be put on pills and their pain would linger. Now those patients can have a treatment that works for them.”
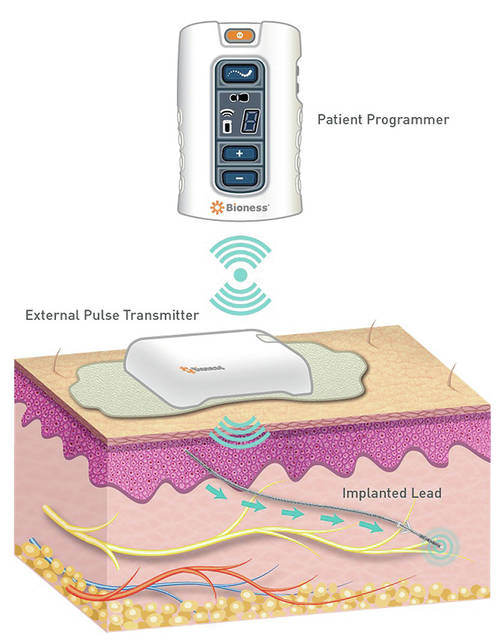
A StimRouter implant requires no hospital stay, no need to go under general anesthesia, and is usually completed in approximately 30 minutes. The treatment is designed to reduce pain by specifically targeting the affected peripheral nerve by delivering gentle stimulating pulses that disrupt pain signals before they reach the brain. StimRouter is intended to be a cost-effective and long-term alternative to prescription opioids. The StimRouter implant procedure requires use of only local anesthesia and is performed with only one to two small incisions. Following surgery, the patient wears a small, externally-worn patch that generates power to the implanted lead. Patients control the system using a wireless, handheld programming device.
“The opioid epidemic continues to impact people across the world in high numbers,” said Todd Cushman, president and CEO of Bioness. “As clinicians and patients continue to look for effective and sustainable ways to treat and manage pain – without the use of opioid medication – it is rewarding to see patients thrive with our StimRouter technology.”
Unlike oral pain medications that circulate throughout a patient’s entire body, the StimRouter targets the precise area where a patient feels pain, without the common prescription drug side effects. Therapies like opioids have a reduced effect over time and are not sustainable long-term.
The StimRouter also does not have to be a permanent therapy. The small lead can be easily removed if the patient decides to pursue a different treatment.
“It’s a minimally-invasive outpatient procedure,” said Malinowski. “The StimRouter can help patients with their recovery when they may not be symptom-free after months of physical therapy. It is a viable alternative to opiates and other treatments.”
He added, “The StimRouter can be used for chronic joint pain. Previously, pain therapies around joints were always challenging because the joint moves in different directions, which would cause the therapy to lose its effectiveness over time. With this device, we can capture chronicle pain in and around joints whether or not the joints have been replaced.”
“We’re happy to be able to provide this safe and effective alternative to get our patients back to the activities they enjoy,” said Porter. “The StimRouter is a durable and elegantly placed device that meets a need for chronic pain patients, who to this point have been underserved.”
Adena’s Interventional Pain Management Department was founded with the goal of helping people return to normal, productive living. Its pain medicine doctors are experts at diagnosing the cause of pain and helping to eliminate it. For more information, visit adena.org/painmanagement or call 740-779-4598.
For more about Adena Health System and its services, visit adena.org or follow us on Facebook, Instagram, or Twitter @adenamedical.
Submitted by Jason Gilham, communications manager, Adena Health System.